The brain is regarded as the most complex structure in our universe, and much is still unknown about this extraordinary organ. Conventional thinking amongst neuroscientists used to be that the brain cells you were born with were fixed and incapable of regeneration, being the sum of brain cells for the rest of your life.
It is only during the 1990’s, with the aid of more advanced technology, that evidence of neural stem cells was found in certain areas in the brains of adult humans. As stem cells have the unique ability to transform into new cells through cell division, it means the brain can grow new brain cells, a process called neurogenesis.

What is neurogenesis?
The term neurogenesis is made up of the words “neuro”, which relates to nerves or the nervous system, and the word “genesis”, which refers to the origin or beginning of something. Neurogenesis therefore refers to the growth and development of new nerve cells in the brain, called neurons. A neuron is a specialized cell that transmits nerve impulses.
While neurogenesis is at its most active while a baby’s neurons are developing in the womb, scientists found that neural stem cells continue to form new neurons in two areas in the adult brain throughout life, firstly in the dentate gyrus area in the hippocampus, which is an area that allows the brain to differentiate between different memories, such as where you have put your keys today as opposed to the day before. The hippocampus is the area in the brain associated with memory and learning.
The second area where new brain cells are formed is in the subventricular zone (SVZ) in the brain, where for example newly formed neurons end up in the olfactory bulb, the center for the sense of smell.
The olfactory bulb receives neural signals about odours detected by smell receptor cells in the nasal cavity. The axons of these smell receptor cells extend directly into the olfactory bulb, where information about odours is processed. While the senses of aging people deteriorate over time, the sense of smell remains stronger in comparison, due to neurogenesis in the subventricular zone.
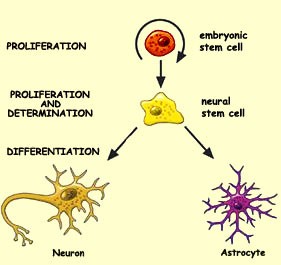
How stem cells form neurons:
Every cell in the human body contains all the genes to form a human body and any given cell acquires its specific nature when only certain genes are activated, for example the genes that are specific to neurons, or skin cells.
Stem cells are regarded as reserve cells with the ability to change into many different types of cells and to grow indefinitely. Stem cells go through three different processes to transform into mature neurons:
- Cell proliferation is the first of these processes, which increases the number of cells. This process starts early, in the first few weeks, in an embryo’s development. After the baby is born, the multiplication of synaptic connections becomes the predominant activity.
- The second process is a critical stage called determination in which the destiny of certain cells is decided, as they become the precursors to various types of neurons and glial cells, for example an astrocyte type of glial cell.
- The third process to form mature neurons is called differentiation, by which a population of neurons gives rise to (differentiate) subpopulations that are specific to the various areas in the nervous system. During this stage neurons migrate to their final destinations, where they make specific connections to other neurons.
Neuron’s functions are quite different from cells in homogeneous populations in the body, for example cells in the pancreas secrete insulin, no matter where in the pancreas the cells are located. The functions of neurons, on the other hand, are determined by their position in the nervous system. A cholinergic neuron, which secrets the neurotransmitter acetylcholine, in the spinal cord has a different function than a cholinergic neuron in the retina of the eye. The location of a neuron determines the connections that it makes with other neurons and the type of neurotransmitter that it secretes.
Stem cells can divide indefinitely to produce more stem cells, or may differentiate to produce more specialized cells, for example neural progenitor cells. These descendants of stem cells can differentiate further to create specialized cell types in the brain such as neurons or glial cells. Glial cells provide supporting and protecting functions to the nervous system and do not produce electrical impulses.
Until a few years ago, scientists believed that glial cells outnumbered neurons by a ratio of 10:1, with 100 billion neurons and one trillion glial cells in the human brain. A new counting method for brain cells, using isotropic fractionator technology, (with results reported as recent as 2017), has however indicated an average of 86 billion neurons and 85 billion non-neural cells in the human brain, which means a ration of not even 1:1 between neurons and glial cells.
The importance of exercise in neurogenesis:
A healthy brain depends on maintaining a healthy network of blood vessels, as the brain is highly dependent on blood flow. The brain constitutes only about 2-3% of body mass, but receives approximately 15% of the body’s entire blood supply. Neurons need a constant supply of oxygen from the bloodstream to function and blood flow increases to regions where neurons become more active. A healthy network of blood vessels is crucial to brain function.
The importance of regular exercise for brain health stems from the improved blood flow to the brain that occurs during exercise, especially aerobic exercise such as walking, running, or cycling.
Regular physical exercise increases the growth of new blood vessels in the brain, which is believed to contribute to the optimal functioning of the brain, as well as preventing loss of overall brain volume (associated with reduced cognitive function) and shrinkage of the hippocampus (associated with loss of memory). Regular exercise is also one of the main contributors to neurogenesis throughout our lifetime.
Exercise also results in various other beneficial responses in the brain, such as an increase in the release of neurotrophic factors in the bloodstream. Neurotrophins are proteins that help to stimulate and control neurogenesis, of which a protein called brain-derived neurotrophic factor (BDNF) is one of the most active of the neurotrophic family of growth factors. BDNF is a neuropeptide that is associated with cognitive improvement, as it regulates axonal growth, neuronal survival, and differentiation of neurons. BDNF also affects synaptic plasticity (plasticity refers to the brain’s ability to change and adapt from experience.) and is linked to adult neurogenesis.
Like most proteins in the body, BDNF is encoded by a gene. The BDNF gene provides the cell’s instructions to make BDNF. Interestingly, the gut microbiota can modulate BDNF function in the central nervous system, for example by changes in the availability and actions of short chain fatty acids in the brain. Certain types of exercise have been shown to substantially (threefold) increase BDNF synthesis in the human brain.
Rodent studies have shown a direct relationship between physical activity and neurogenesis in the hippocampus, such as improvement in neurogenesis and enhanced synaptic plasticity that correlates with a significant increase in hippocampus-dependent learning and memory tasks.
Interestingly, studies in rodents as well as human studies have shown deficits in hippocampus functioning due to prenatal alcohol exposure.
Neurogenesis and aging:
The rise in life expectancy in modern society has resulted in intensified efforts to find new approaches to improve age-related brain dysfunction. Normal aging is characterized by changes in brain structure and cognitive weakening due to, amongst others, an irreversible loss of neurons, while the hippocampus typically shrinks by 1% to 2% a year in older adults. Progressive memory impairment is part and parcel of aging and scientists are of the opinion that reduced neurogenesis and synaptic plasticity play a role.
Although human autopsies found evidence that neurogenesis still occurs into old age, (experimental and ethical constraints preventing human studies), rodent studies have shown that the forming of new neurons gets impaired with advancing age, resulting in a decrease in the numbers of newly generated neurons. Scientists found that the number of newborn neurons in older rats are only about 10% of the numbers detectable in young rats. The decrease in newborn neurons in the aged hippocampus could also be correlated with a reduction in the division rate of neural stem cells, which becomes gradually exhausted over time.
Studies suggests that exercise may counteract the cognitive decline that occurs in late adulthood in humans, while animal studies have indicated that exercise improves neurogenesis and learning in aged mice.
While studies have shown that physical activity may have a positive effect on brain function throughout the lifespan, it is unknown whether exercise that begun late in life can arrest the cognitive decline that is associated with aging.
The shortening of telomeres and its effect on cell division is another biological process that plays a role in the aging of cells in the body. Within a single cell the long strings – like shoelaces – indicate the chromosomes (DNA), which have protective ends called telomeres that prevents the chromosomes from disintegrating – like the protective plastic tips at the ends of shoelaces. When cell division occurs and everything in the cell is split in two and two new cells are formed, the telomeres become slightly shorter, and this shortening of the telomeres occurs each time a cell divides. This is significant as you age, as there are dire consequences when the telomeres run out and such cells are now unable to further multiply or offer protection. Recent studies suggests that the telomeres of neurons remain stable throughout life, while the telomeres in glial cells become significantly shorter with aging.
Neurogenesis and lifestyle:
Apart from the vital role of daily exercise, recent science shows that other lifestyle factors can also affect the rate of neurogenesis. Certain lifestyle factors are deemed to boost neurogenesis, such as:
- An enriched environment with new experiences and challenges, for example doing activities such as travelling, learning a new skill or new language, and taking on intellectual challenges not only trigger neurogenesis, but it also means those new brain cells are likely to stay long term. An enriched environment means getting out of your comfort zone, experiencing new things, and being mentally challenged.
- Studies have shown that meditation in the form of yoga can benefit the body and the mind, while it may support neurogenesis through increased hippocampus volume and increased grey matter (neural) density.
- Diet and eating healthy fats are key to brain health, as fats make up 60% of the raw material in the brain, with Omega 3 playing an important part in neurogenesis.
- There are indications that temporary fasting and the restriction of calories may trigger the increased growth of neurons.
Conclusion:
Neuroscience provides proof that the brain continues to make new brain cells. It is up to us to enhance this process through regular physical activity, healthy eating, and new mental and physical challenges, in support of healthy brain function for as long as possible.
References:
Can you grow new brain cells? Published 14 September 2016. Harvard Medical School. (www.health.harvard.edu)
What is neurogenesis? Published online. Queensland Brain Institute. The University of Queensland, Australia. (www.qbi.uq.edu.au)
Exercise and the brain: three ways physical activity changes its very structure. Published 17 November 2020. The Conversation. (www.theconversation.com)
The long run: neuroprotective effects of physical exercise on adult neurogenesis from youth to old age. Published May 2017. National Center for Biotechnology Information. National Library of Medicine. National Institutes of Health. USA. (www.nbci.nlm.nih.gov)
5 ways to boost your neurogenesis. Published 6 December 2018. NeuroTrackerX. (www.neurotrackerx.com)
What is neurogenesis? Published online and updated 27 February 2019. News. Medical Lifesciences. (www.news-medical.net)
Researchers discover a mechanism linked to stem cell aging. Published online and reviewed 24 February 2021. News. Medical Lifesciences. (www.news-medical.net)
The search for true numbers of neurons and glial cells in the human brain: a review of 150 years of cell counting. Published December 2016. PMC. National Center for Biotechnology Information. National Library of Medicine. National Institutes of Health. USA. (www.nbci.nlm.nih.gov)
Changes in telomere length with aging in human neurons and glial cells revealed by quantitative fluorescence in situ hybridization analysis. Published 10 August 2018. PubMed. National Center for Biotechnology Information. National Library of Medicine. National Institutes of Health. USA. (www.nbci.nlm.nih.gov)
HEALTH INSIGHT
